
Publications

19. Overcrowded Alkene Photo-Redox-Switches Based on Quinolinium/Carbene Building Blocks
C. Burdenski, P. W. Antoni, M. E. Baumert, L. Ziemann, S. C. Sau, J. J. Holstein, M. Castro, N. Kumar, O. Filiba, I. Schapiro, M. M. Hansmann
Angew. Chem. Int. Ed.2026, e9975231
-
The design and characterization of a novel class of overcrowded alkene-based photo/redox switches are presented. Folded and twisted tetrasubstituted alkenes are prepared in a modular fashion by the combination of quinolinium salts and stable carbenes. The compounds show multi-stimuli responsive properties: besides electrochemical redox-switching, which allows C─C bond rotation via a two-electron redox process, non-symmetrical compounds enable photochemically driven E/Z-switching. The different oxidation states can be isolated and fully characterized including structural changes verified by x-ray diffraction. Excellent photostability for E/Z-switching was observed in selected cases and is supported by UV–vis, NMR, and x-ray data as well as computations. TD-DFT calculations accurately predict the absorption properties of the E/Z-photo-switches and the substituent effects governing photo-stationary states and half-lifes. While the thermal E/Z-back-switching proceeds via a rather high energy barrier featuring a diradical transition state, electrochemical switching via the radical cation oxidation state allows instantaneous E/Z-isomerization, which is catalytic in electrons (electron hole catalysis). The mechanism of the switching process is supported in detail by quantum chemical calculations including nonadiabatic molecular dynamics simulations.
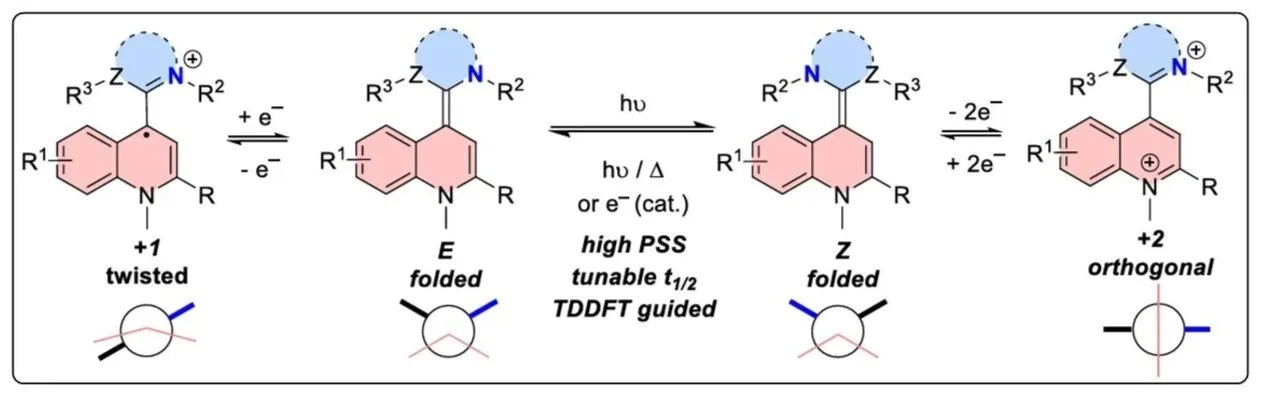

18. Dicationic Acridinium/Carbene Hybrids as Strongly Oxidizing Photocatalysts
S. C. Sau, M. Schmitz, C. Burdenski, M. Baumert, P. W. Antoni, C. Kerzig, M. M. Hansmann
J. Am. Chem. Soc. 2024, 146, 3416–3426.
-
A new design concept for organic, strongly oxidizing photocatalysts is described based upon dicationic acridinium/carbene hybrids. A highly modular synthesis of such hybrids is presented, and the dications are utilized as novel, tailor-made photoredox catalysts in the direct oxidative C–N coupling. Under optimized conditions, benzene and even electron-deficient arenes can be oxidized and coupled with a range of N-heterocycles in high to excellent yields with a single low-energy photon per catalytic turnover, while commonly used acridinium photocatalysts are not able to perform the challenging oxidation step. In contrast to traditional photocatalysts, the hybrid photocatalysts reported here feature a reversible two-electron redox system with regular or inverted redox potentials for the two-electron transfer. The different oxidation states could be isolated and structurally characterized supported by NMR, EPR, and X-ray analysis. Mechanistic experiments employing time-resolved emission and transient absorption spectroscopy unambiguously reveal the outstanding excited-state potential of our best-performing catalyst (+2.5 V vs SCE), and they provide evidence for mechanistic key steps and intermediates.


17. Stable Abnormal N-Heterocyclic Carbenes and their Applications
S. C. Sau, P. K. Hota, S. K. Mandal, M. Soleilhavoup, G. Bertrand
Chem. Soc. Rev. 2020, 49, 1233–1252.
-
Although N-heterocyclic carbenes (NHCs) have been known as ligands for organometallic complexes since the 1960s, these carbenes did not attract considerable attention until Arduengo et al. reported the isolation of a metal-free imidazol-2-ylidene in 1991. In 2001 Crabtree et al. reported a few complexes featuring an NHC isomer, namely an imidazol-5-ylidene, also termed abnormal NHC (aNHCs). In 2009, it was shown that providing to protect the C-2 position of an imidazolium salt, the deprotonation occurred at the C-5 position, affording imidazol-5-ylidenes that could be isolated. Over the last ten years, stable aNHCs have been used for designing a range of catalysts employing Pd(II), Cu(I), Ni(II), Fe(0), Zn(II), Ag(I), and Au(I/III) metal based precursors. These catalysts were utilized for different organic transformations such as the Suzuki–Miyaura cross-coupling reaction, C–H bond activation, dehydrogenative coupling, Huisgen 1,3-dipolar cycloaddition (click reaction), hydroheteroarylation, hydrosilylation reaction and migratory insertion of carbenes. Main-group metal complexes were also synthesized, including K(I), Al(III), Zn(II), Sn(II), Ge(II), and Si(II/IV). Among them, K(I), Al(III), and Zn(II) complexes were used for the polymerization of caprolactone and rac-lactide at room temperature. In addition, based on the superior nucleophilicity of aNHCs, relative to that of their nNHCs isomers, they were used for small molecules activation, such as carbon dioxide (CO2), nitrous oxide (N2O), tetrahydrofuran (THF), tetrahydrothiophene and 9-borabicyclo[3.3.1]nonane (9BBN). aNHCs have also been shown to be efficient metal-free catalysts for ring opening polymerization of different cyclic esters at room temperature; they are among the most active metal-free catalysts for ε-caprolactone polymerization. Recently, aNHCs successfully accomplished the metal-free catalytic formylation of amides using CO2 and the catalytic reduction of carbon dioxide, including atmospheric CO2, into methanol, under ambient conditions. Although other transition metal complexes featuring aNHCs as ligand have been prepared and used in catalysis, this review article summarize the results obtained with the isolated aNHCs.


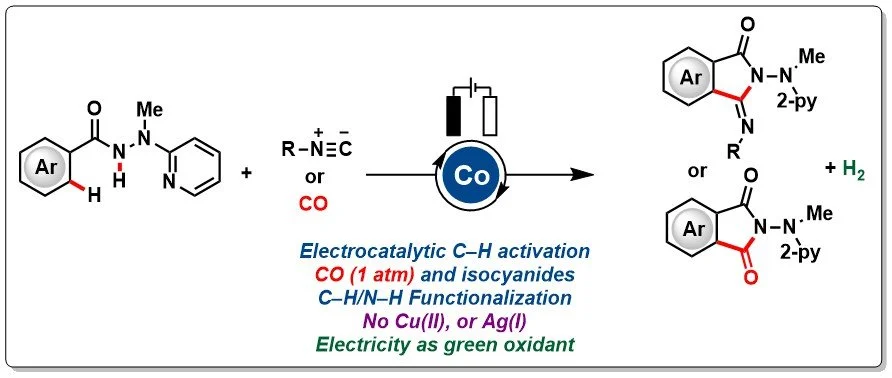
16. Cobaltaelectro Catalyzed C−H Activation with Carbon Monoxide or Isocyanides
S. C. Sau, P. K. Hota, S. K. Mandal, M. Soleilhavoup, G. Bertrand
ChemSusChem 2019, 12, 3023–3027.
-
Electrochemical oxidative C−H/N−H activations with isocyanides have been realized with a versatile cobalt catalyst. The widely applicable cobalt catalysis manifold further enabled electrooxidative C−H/N−H carbonylations with carbon monoxide under ambient conditions. The C−H functionalizations were efficiently realized with ample scope and outstanding functional group tolerance in a user-friendly undivided cell setup.

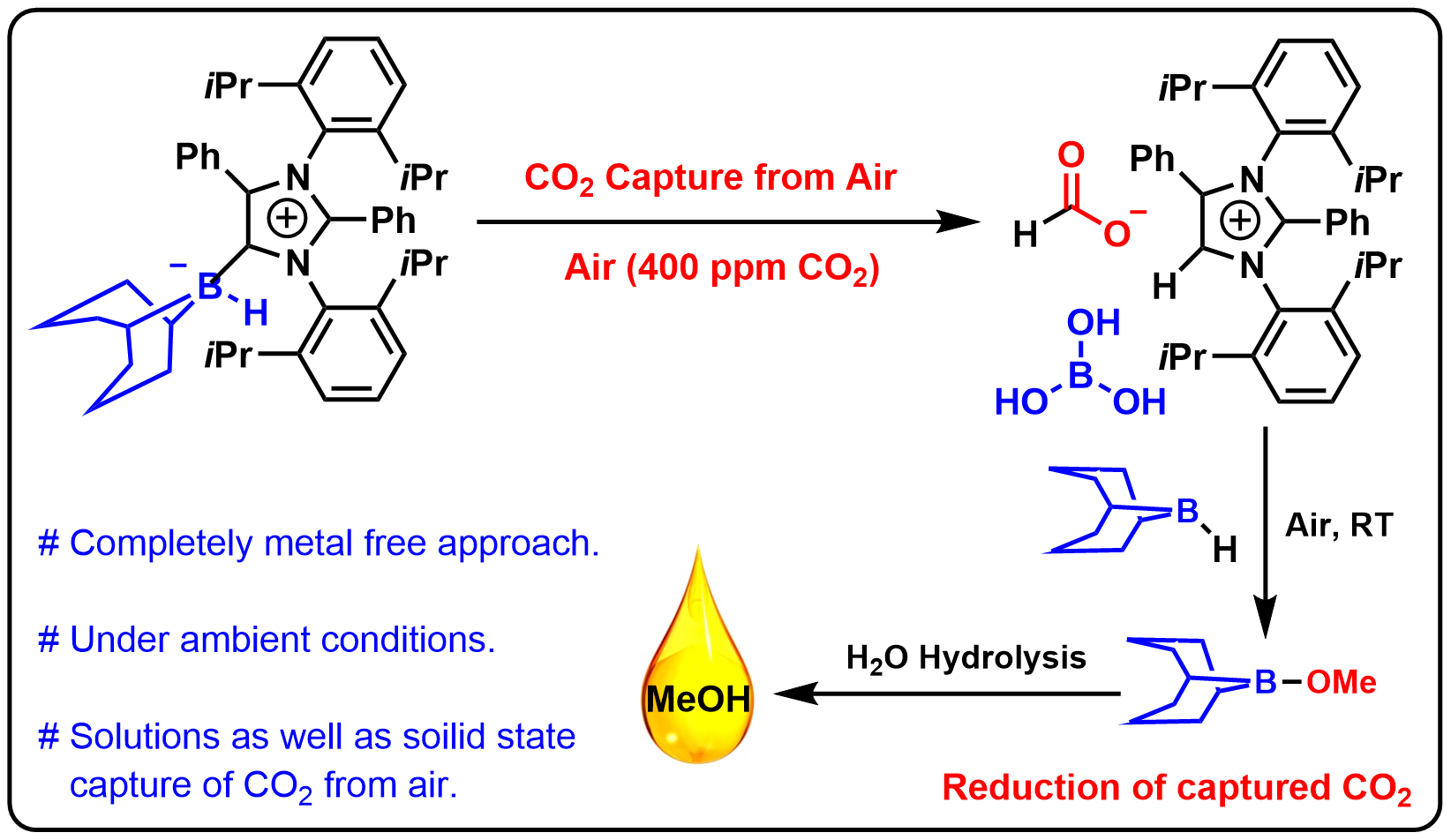
15. Transforming Atmospheric CO2 into Alternative Fuels: A Metal-Free Approach under Ambient Conditions
S. C. Sau, R. Bhattacharjee, P. K. Hota, P. K. Vardhanapu, G. Vijaykumar, R. Govindarajan, A. Datta, S. K. Mandal
Chem. Sci. 2019, 10, 1879–1884.
-
This work demonstrates the first-ever completely metal-free approach to the capture of CO2 from air followed by reduction to methoxyborane (which produces methanol on hydrolysis) or sodium formate (which produces formic acid on hydrolysis) under ambient conditions. This was accomplished using an abnormal N-heterocyclic carbene (aNHC)–borane adduct. The intermediate involved in CO2 capture (aNHC-H, HCOO, B(OH)3) was structurally characterized by single-crystal X-ray diffraction. Interestingly, the captured CO2 can be released by heating the intermediate, or by passing this compound through an ion-exchange resin. The capture of CO2 from air can even proceed in the solid state via the formation of a bicarbonate complex (aNHC-H, HCO3, B(OH)3), which was also structurally characterized. A detailed mechanism for this process is proposed based on tandem density functional theory calculations and experiments.

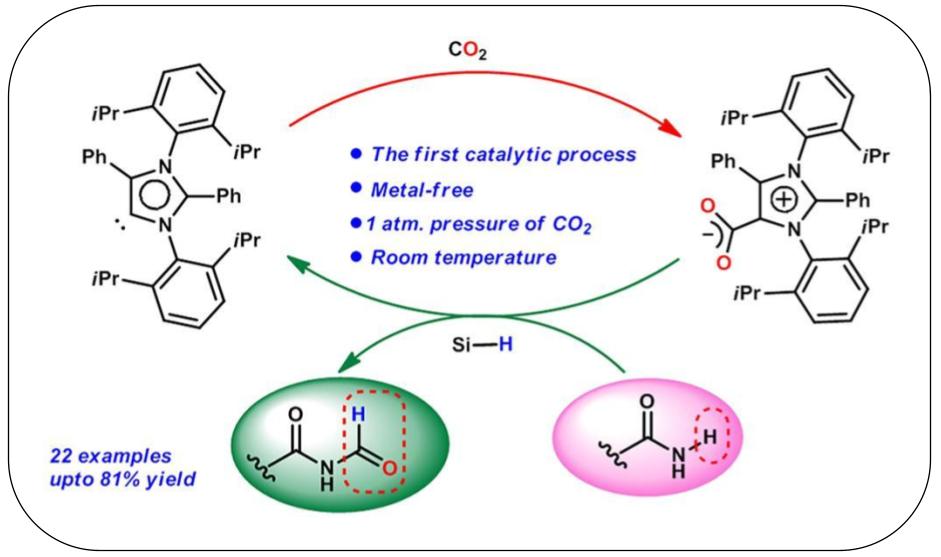
14. Metal-Free Catalytic Formylation of Amides Using CO2 under Ambient Conditions
P. K. Hota, S. C. Sau, S. K. Mandal
ACS Catal. 2018, 8, 11999−12003.
-
This study reports the metal-free formylation of amides using carbon dioxide under ambient conditions. An abnormal N-heterocyclic carbene (aNHC) acts as an efficient catalyst for the formylation of amides in the presence of hydrosilane at room temperature. This methodology enables the formation of a C–N bond and can be utilized in building up core moieties of two natural products having strong larvicidal activity such as alatamide and lansiumamide A. A preliminary mechanistic picture for this transformation has been proposed through isolation of reaction byproduct (confirmed by single crystal X-ray study) as well as by characterizing intermediates with spectroscopy

13. Electrooxidative Allene Annulations by Mild Cobalt-Catalyzed C−H Activation
P. K. Hota, S. C. Sau, S. K. Mandal
ACS Catal. 2018, 8, 9140−9147.
-
Versatile cobalt catalysis enabled electrochemical C–H activation with allenes. Thus, allene annulations were accomplished in terms of C–H/N–H functionalizations with excellent levels of chemoselectivity, site selectivity, and regioselectivity under exceedingly mild conditions. Detailed mechanistic studies were conducted, including reactions with isotopically labeled compounds, kinetic investigations, and in-operando infrared spectroscopic studies. Further, computational studies were supportive of a non-rate-determining C–H cleavage and gave key insights into the regioselectivity of the allene annulation. The practical utility of the user-friendly approach was furthermore highlighted by gram-scale electrocatalysis.
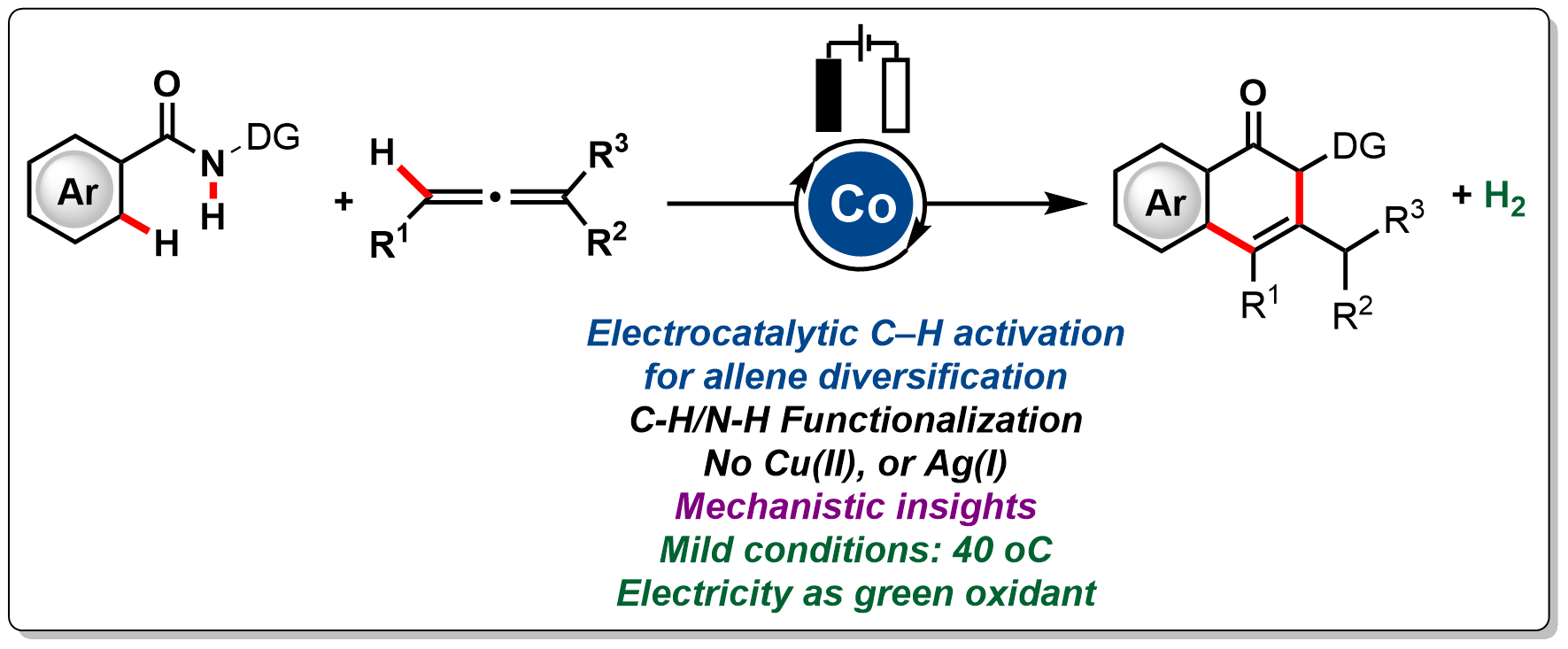

12. Halo-Bridged Abnormal NHC Palladium(II) Dimer for Catalytic Dehydrogenative Cross-Coupling Reactions of Heteroarenes
P. Sreejyothi,≠ S. C. Sau,≠ P. K. Vardhanapu, S. K. Mandal (≠Authors contributed equally)
J. Org. Chem. 2018, 83, 9403−9411.
-
This work describes the dehydrogenative coupling of heteroarenes using a dimeric halo-bridged palladium(II) catalyst bearing an abnormal NHC (aNHC) backbone. The catalyst can successfully activate the C–H bond of a wide range of heteroarenes, which include benzothiazole, benzoxazole, thiophene, furan, and N-methylbenzimidazole. Further, it exhibited good activity for heteroarenes bearing various functional groups such as CN, CHO, Me, OMe, OAc, and Cl. Additionally, we isolated the active catalyst by performing stoichiometric reaction and characterized it as the acetato-bridged dimer of (aNHC)PdOAc by single-crystal X-ray study.
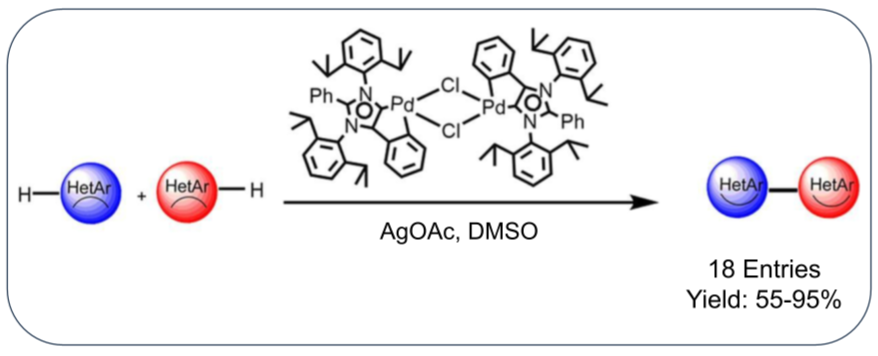

11. Exploring Closed Shell Cationic Phenalenyl: From Catalysis to Spin Electronics
A. Mukherjee, S. C. Sau, S. K. Mandal
Acc. Chem. Res. 2017, 50, 1679−1691.
-
The odd alternant hydrocarbon phenalenyl (PLY) can exist in three different forms, a closed-shell cation, an open-shell radical, and a closed-shell anion, using its nonbonding molecular orbital (NBMO). The chemistry of PLY-based molecules began more than five decades ago, and so far, the progress has mainly involved the open-shell neutral radical state. Over the last two decades, we have witnessed the evolution of a range of PLY-based radicals generating an array of multifunctional materials. However, it has been admitted that the practical applications of PLY radicals are greatly challenged by the low stability of the open-shell (radical) state.
Recently, we took a different route to establish the utility of these PLY molecules using the closed-shell cationic state. In such a design, the closed-shell unit of PLY can readily accept free electrons, stabilizing in its NBMO upon generation of the open-shell state of the molecule. Thus, one can synthetically avoid the unstable open-shell state but still take advantage of this state by in situ generating the radical through external electron transfer or spin injection into the empty NBMO. It is worth noting that such approaches using closed-shell phenalenyl have been missing in the literature.
This Account focuses on our recent developments using the closed-shell cationic state of the PLY molecule and its application in broad multidisciplinary areas spanning from catalysis to spin electronics. We describe how this concept has been utilized to develop a variety of homogeneous catalysts. For example, this concept was used in designing an iron(III) PLY-based electrocatalyst for a single-compartment H2O2 fuel cell, which delivered the best electrocatalytic activity among previously reported iron complexes, organometallic catalysts for various homogeneous organic transformations (hydroamination and polymerization), an organic Lewis acid catalyst for the ring opening of epoxides, and transition-metal-free C–H functionalization catalysts. Moreover, this concept of using the empty NBMO present in the closed-shell cationic state of the PLY moiety to capture electron(s) was further extended to an entirely different area of spin electronics to design a PLY-based spin-memory device, which worked by a spin-filtration mechanism using an organozinc compound based on a PLY backbone deposited over a ferromagnetic substrate.
In this Account, we summarize our recent efforts to understand how this unexplored closed-shell state of the phenalenyl molecule, which has been known for over five decades, can be utilized in devising an array of materials that not only are important from an organometallic chemistry or organic chemistry point of view but also provide new understanding for device physics.
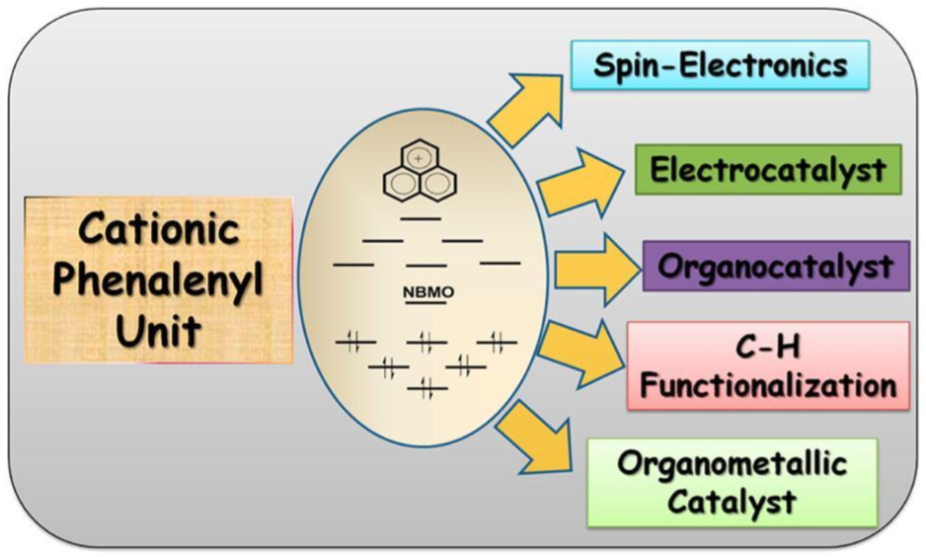

10. Direct C-H Arylation of Heteroarenes with Aryl Chlorides by Using an Abnormal N-Heterocyclic Carbene Palladium Catalyst
J. Ahmed,≠ S. C. Sau,≠ P. Sreejyothi, P. K. Hota, P. K. Vardhanapu, G. Vijaykumar, S. K. Mandal (≠Authors contributed equally)
Eur. J. Org. Chem. 2017, 2017, 1004–1011.
-
Herein, we report a versatile catalytic system for the direct C–H arylation of heteroarenes with activated aryl chloride substrates. The catalyst works successfully for a variety of heteroarenes and aryl chloride coupling partners under very low catalyst-loading conditions. We have successfully performed the direct C–H arylations of 1-methylpyrrole, 1-methylindole, furan, thiophene, furfural, and N-benzyl-1,2,3-triazole with aryl chloride partners in good yields without the use of any additives. Furthermore, we used this catalytic process to develop a one-pot synthetic protocol for the muscle relaxant dantrolene on a gram scale. Additionally, the present catalytic system can be used to perform consecutive arylations in one pot.

9. Metal-Free Reduction of CO2 to Methoxyborane under Ambient Conditions through Borondiformate Formation
S. C. Sau, R. Bhattacharjee, P. K. Vardhanapu, G. Vijaykumar, A. Datta, S. K. Mandal
Angew. Chem. Int. Ed. 2016, 55, 15147–15151.
-
An abnormal N-heterocyclic carbene (aNHC) based homogeneous catalyst has been used for the reduction of carbon dioxide to methoxyborane in the presence of a range of hydroboranes under ambient conditions and resulted in the highest turnover number of 6000. A catalytically active reaction intermediate, [aNHC-H⋅9BBN(OCOH)2] was structurally characterized and authenticated by NMR spectroscopy. A detailed mechanistic cycle of this catalytic process via borondiformate formation has been proposed from tandem experimental and computational experiments.
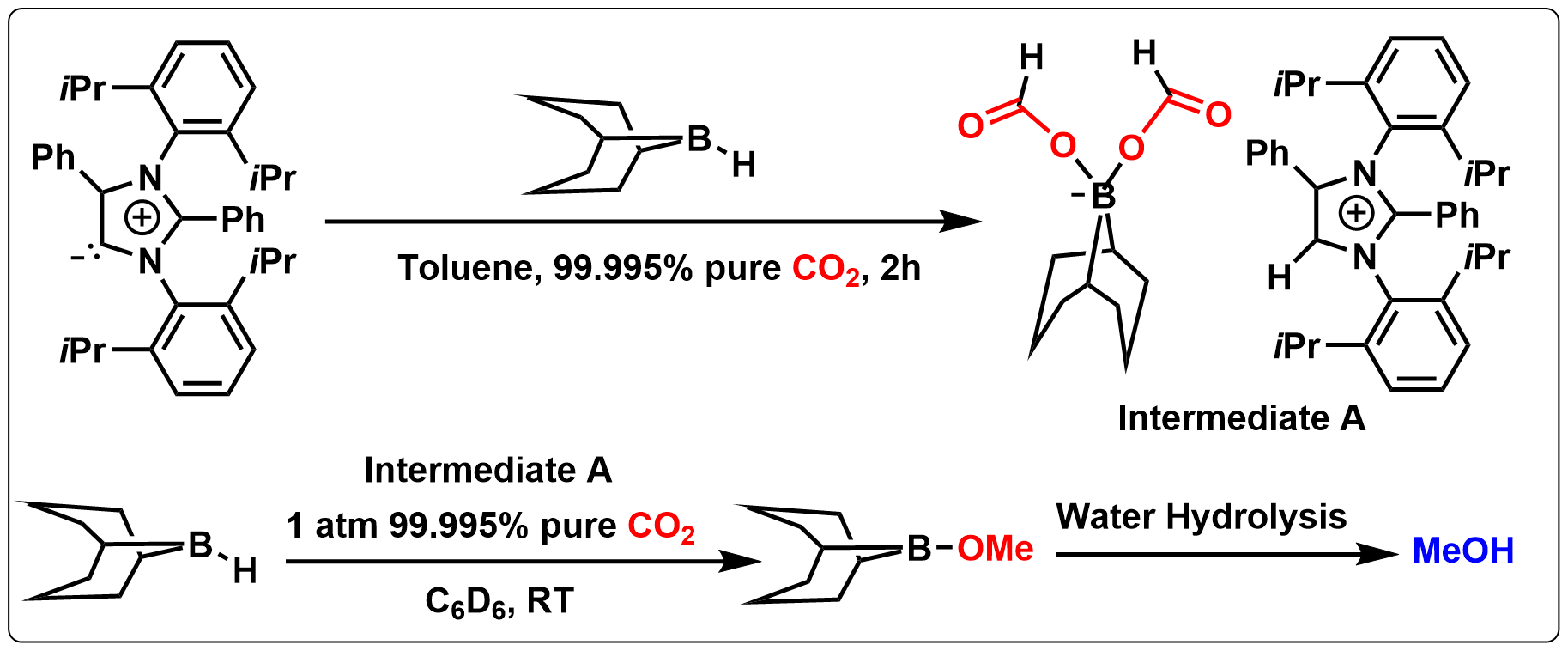

8. Open Shell Phenalenyl in Transition Metal-Free Catalytic C-H Functionalization
R. Paira, B. Singh, P. K. Hota, J. Ahmed, S. C. Sau, J. P. Johnpeter, S. K. Mandal
J. Org. Chem. 2016, 81, 2432–2441.
-
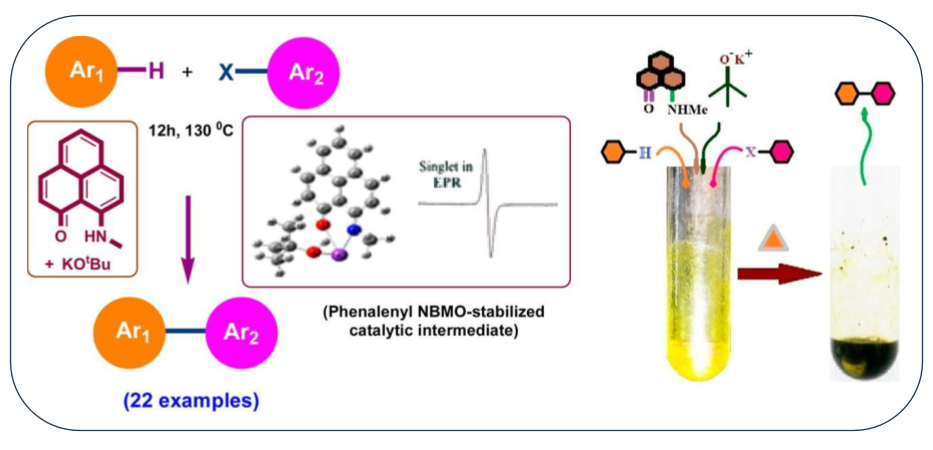
Open-shell phenalenyl chemistry has widely been explored in the last five decades demonstrating its potential in various applications including molecular switch, spin memory device, molecular battery, cathode material, etc. In this article, we have explored another new direction of open-shell phenalenyl chemistry toward transition metal-free catalytic C–H functionalization process. A phenalenyl ligand, namely, 9-methylamino-phenalen-1-one (4a), promoted chelation-assisted single electron transfer (SET) process, which facilitates the C–H functionalization of unactivated arenes to form the biaryl products. The present methodology offers a diverse substrate scope, which can be operated without employing any dry or inert conditions and under truly transition metal based catalyst like loading yet avoiding any expensive or toxic transition metal. This not only is the first report on the application of phenalenyl chemistry in C–H functionalization process but also provides a low-catalyst loading organocatalytic system (up to 0.5 mol % catalyst loading) as compared to the existing ones (mostly 20–40 mol %), which has taken advantage of long known phenalenyl based radical stability through the presence of its low-lying nonbonding molecular orbital.

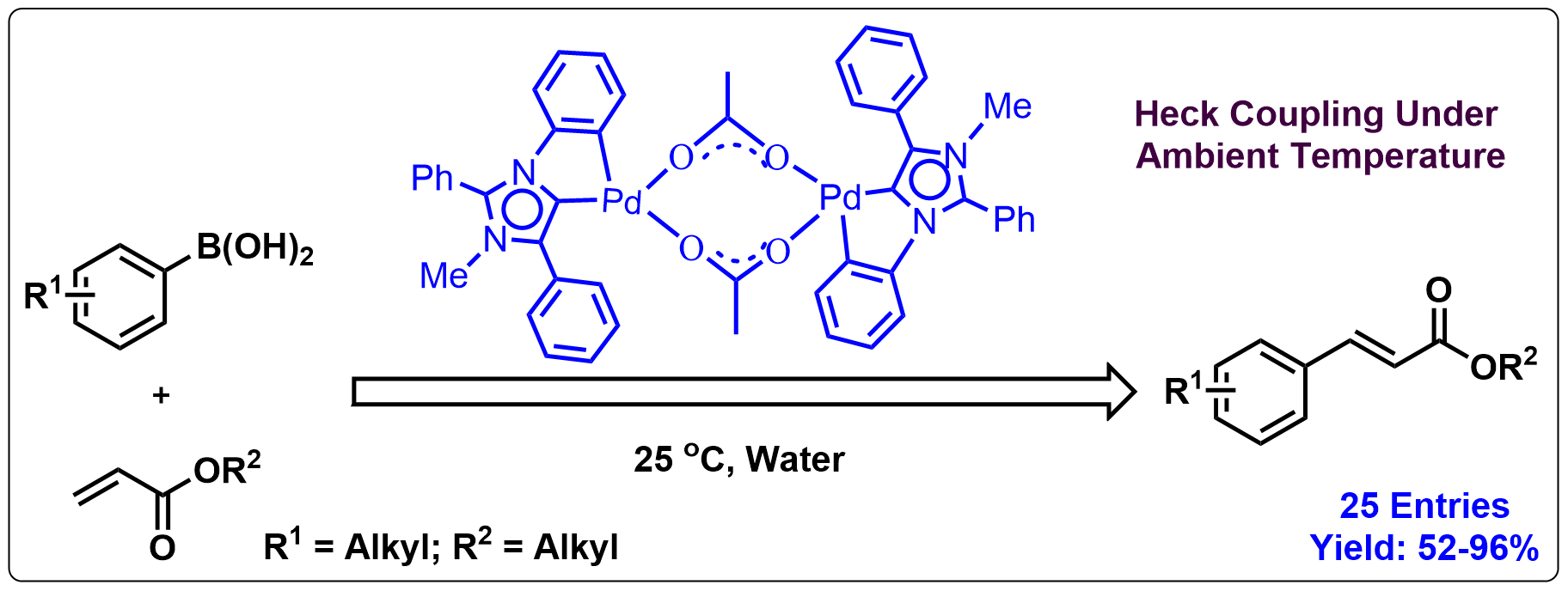
7. An Abnormal N-Heterocyclic Carbene Based Palladium Dimer: Aqueous Oxidative Heck Coupling Under Ambient Temperature
P. K. Hota, G. Vijaykumar, A. Pariyar, S. C. Sau, T. K. Sen, S. K. Mandal
Adv. Synth. Catal. 2015, 357, 3162–3170.
-
Herein we report the synthesis of a new abnormal N-heterocyclic carbene (aNHC)-based ligand and its use in synthesizing an acetate bridged CH activated palladium dimer (1). In complex 1, the carbene ligand displays an abnormal mode of binding with the palladium(II) ion. We used complex 1 as a catalyst for the oxidative Heck coupling reaction of arylboronic acids with electron-deficient as well as electron-rich olefins at ambient temperature in water to produce selectively the β-arylated products in good to excellent yields.

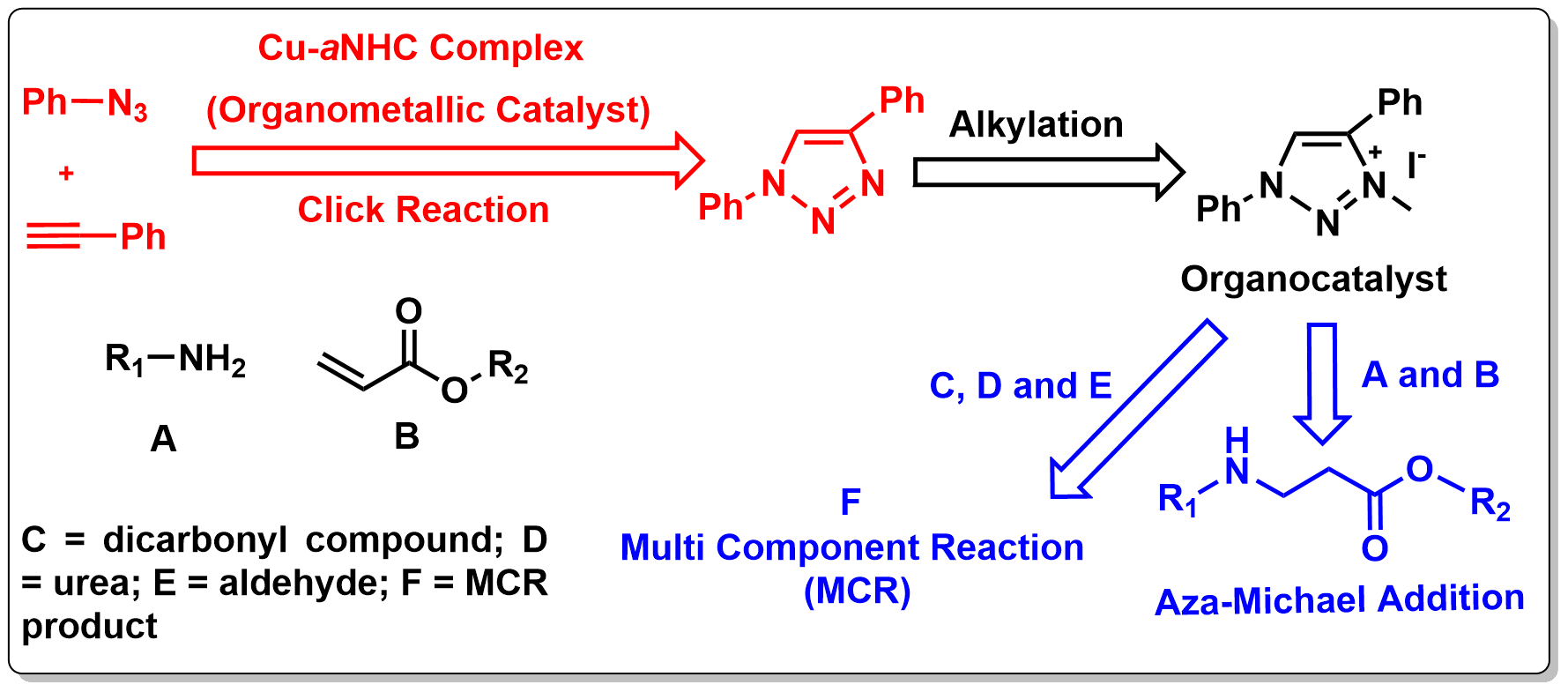
6. One Pot Consecutive Catalysis by Integrating Organometallic Catalysis with Organocatalysis
S. C. Sau, S. Raha Roy, S. K. Mandal
Chem. Asian J. 2014, 9, 2806–2813.
-
The present study integrates two types of catalysis, namely, organometallic catalysis and organocatalysis in one reaction pot. In this process, the product of the first catalytic cycle acts as catalytic component for next catalytic cycle. The abnormal N-heterocyclic carbene–copper-based organometallic catalyst acts as an efficient catalyst for a click reaction to provide triazole, which, in turn, acts as an efficient organocatalyst for different organic transformations, for example, aza-Michael addition and multicomponent reactions, in a consecutive fashion in the same reaction pot.

5. Chemoselective Reduction of the Carbonyl Functionality through Hydrosilylation: Integrating Click Catalysis with Hydrosilylation in One Pot
S. Raha Roy, S. C. Sau, S. K. Mandal
J. Org. Chem. 2014, 79, 9150−9160.
-
Herein we report the chemoselective reduction of the carbonyl functionality via hydrosilylation using a copper(I) catalyst bearing the abnormal N-heterocyclic carbene 1 with low (0.25 mol %) catalyst loading at ambient temperature in excellent yield within a very short reaction time. The hydrosilylation reaction of α,β-unsaturated carbonyl compounds takes place selectively toward 1,2-addition (C═O) to yield the corresponding allyl alcohols in good yields. Moreover, when two reducible functional groups such as imine and ketone groups are present in the same molecule, this catalyst selectively reduces the ketone functionality. Further, 1 was used in a consecutive fashion by combining the Huisgen cycloaddition and hydrosilylation reactions in one pot, yielding a range of functionalized triazole substituted alcohols in excellent yields
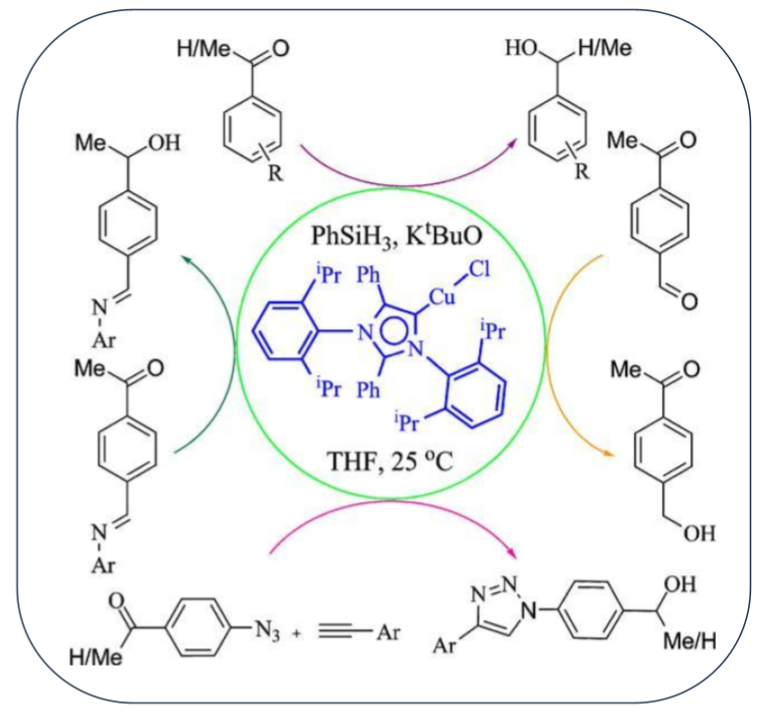

4. An Abnormal N-Heterocyclic Carb-ene Copper(I) Complex in Click Chemistry
S. C. Sau, S. Raha Roy, T. K. Sen, D. Mullangi, S. K. Mandal
Adv. Synth. Catal. 2013, 355, 2982–2991.
-
Herein we report the synthesis of a copper(I) chloro complex using an abnormal N-heterocyclic carbene (aNHC) salt, 1,3-bis(2,6-diisopropylphenyl)-2,4-diphenylimidazolium. The CuCl(aNHC) complex efficiently catalyzed Huisgen 1,3-dipolar cycloaddition reactions (click reactions) of azides with alkynes to give 1,4-substituted 1,2,3-triazoles in excellent yields at room temperature within short reaction time under solvent-free conditions. The catalyst successfully activated benzyl azide and phenylacetylene under the low catalyst loading of 0.005 mol% resulting in a nearly quantitative yield of the product at room temperature with the high TON value of 19,800. The catalyst also exhibits high efficiency in the reaction between sterically hindered azides and alkynes under solvent-free conditions at room temperature. Furthermore, a number of internal alkynes was successfully tested in this copper-catalyzed cycloaddition reaction for synthesis of 4,5-disubstituted triazoles.
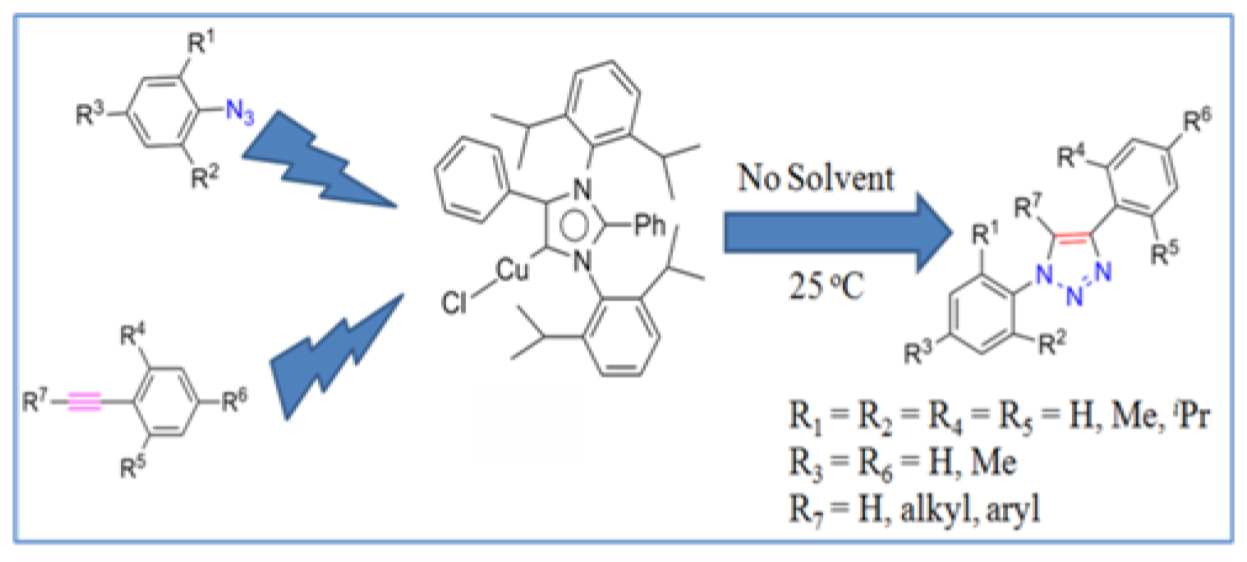

3. Abnormal N-Heterocyclic Carbene Main Group Organometallic Chemistry: A Debut to the Homogeneous Catalysis
T. K. Sen, S. C. Sau, A. Mukherjee, P. K. Hota, S. K. Mandal, B. Maity, D. Koley
Dalton Trans. 2013, 42, 14253–14260.
-
Abnormal N-heterocyclic carbene (aNHC) adducts of zinc(II) (1) and aluminum(III) (2) were synthesized. The compounds were characterized by NMR spectroscopy and elemental analysis. The solid state structures of these complexes (1 and 2) were determined by single crystal X-ray study. Furthermore, these organozinc and organoaluminum adducts (1 and 2) were tested for the ring opening polymerization of cyclic esters. These adducts were found to be quite efficient catalysts for the polymerization of cyclicesters such as rac-lactide (rac-LA), ε-caprolactone (ε-CL), and δ-valerolactone (δ-VL). Furthermore, aNHC zinc adduct has been used as catalyst for the synthesis of a tri-block copolymer.
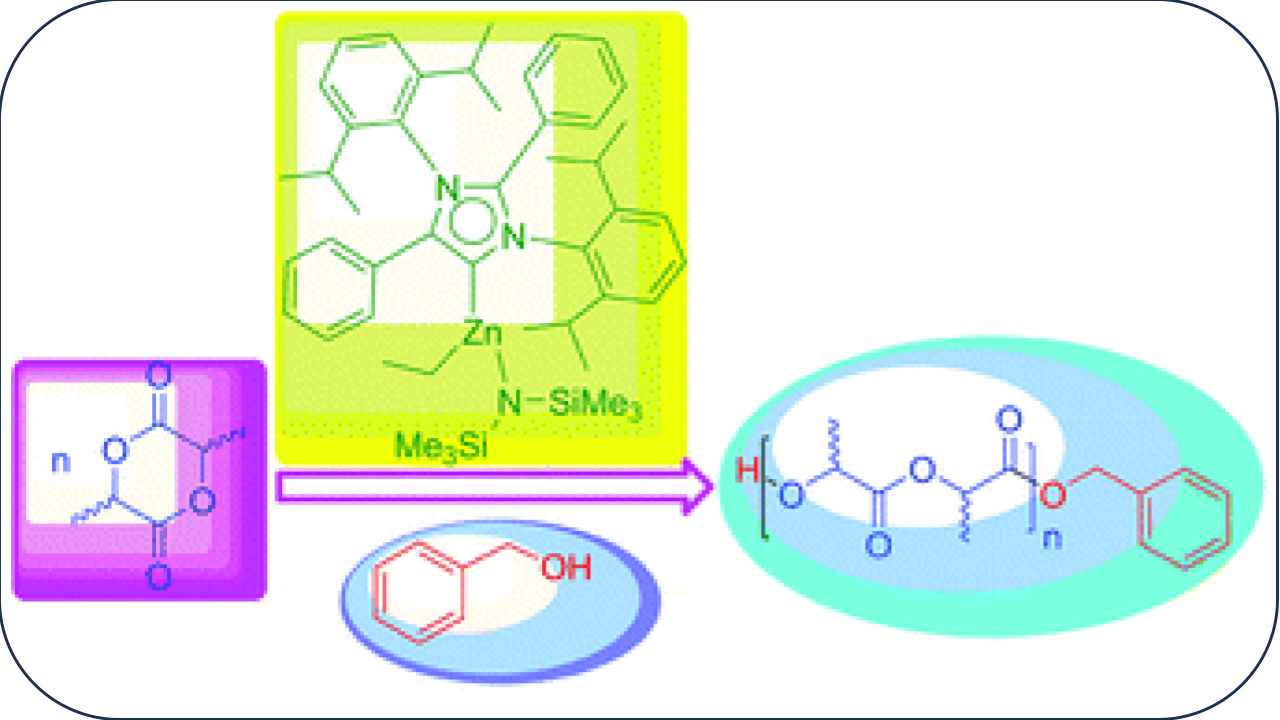

2. Abnormal N-Heterocyclic Carbene Palladium Complex: Living Catalyst for Activation of Aryl Chlorides in Suzuki–Miyaura Cross Coupling
S. C. Sau, S. Santra, T. K. Sen, S. K. Mandal, D. Koley
Chem. Commun. 2012, 48, 555–557.
-
Palladium complexes bearing abnormal N-heterocyclic carbene were used as catalysts in Suzuki–Miyaura cross coupling of aryl chlorides at 25 °C. The catalyst remained active for 10 successive catalytic runs and can activate 4-chlorotoluene at 25 °C with 0.01 mol% catalyst loading resulting in a TON of 9500 within 6 h.


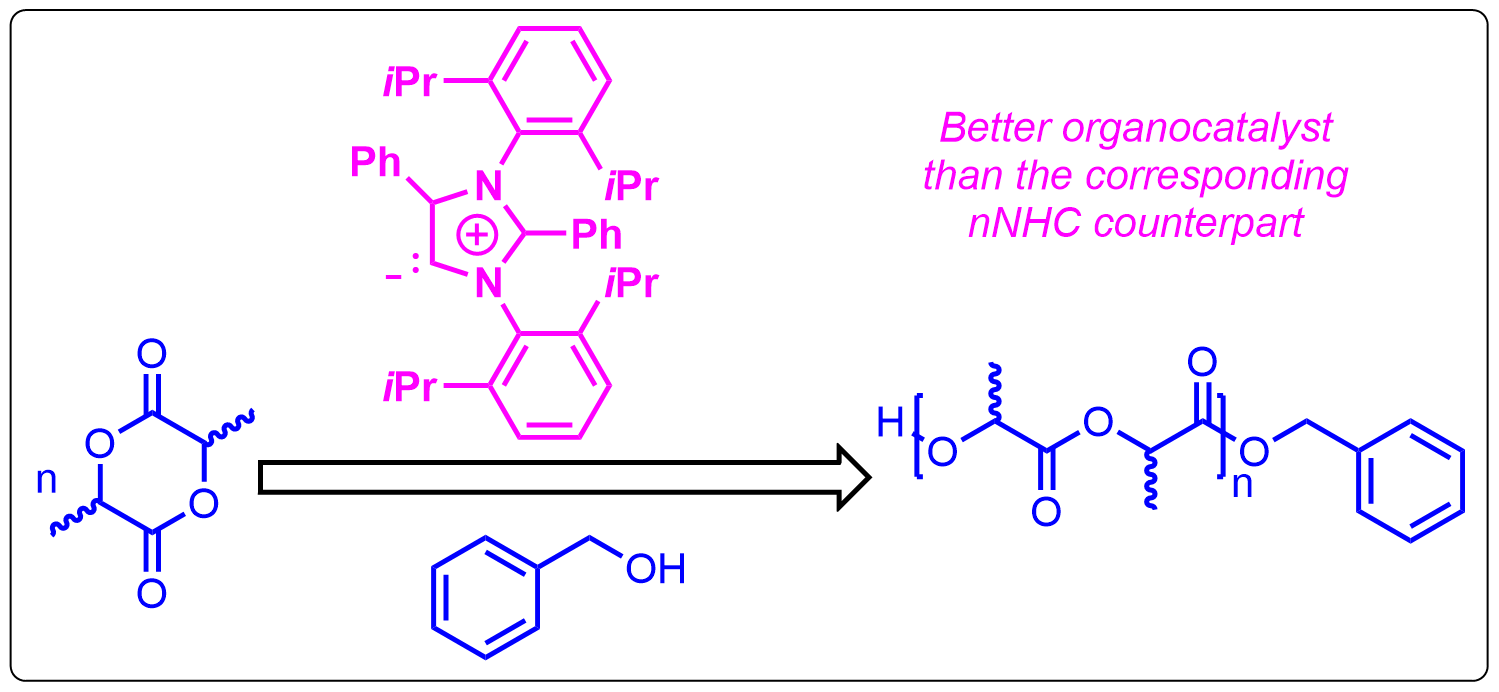
1. Introduction of Abnormal N-Heterocyclic Carbene as an Efficient Organocatalyst: Ring Opening Polymerization of Cyclic Esters
T. K. Sen, S. C. Sau, A. Mukherjee, A. Modak, S. K. Mandal, D. Koley
Chem. Commun. 2011, 47, 11972–11974.
-
The recently isolated abnormal N-heterocyclic carbene (aNHC) has been established as an efficient organocatalyst in ring opening polymerization of three different cyclic estersrac-lactide (rac-LA), ε-caprolactone (ε-CL), and δ-valerolactone (δ-VL). Preliminary DFT calculations indicate that aNHC can be a better organocatalyst than the corresponding nNHC counterpart.
N.B. The journal images shown above were obtained from the respective publications associated with the issue. They are included solely to enhance the visual presentation of this webpage and are not intended for any scientific or research-related use.